eazy
VIP Member
- Aug 30, 2022
- 583
- 907
Why Route of Administration Doesn’t Reduce Hepatotoxic Risk in 17α-Alkylated Anabolic Steroids
By Type-IIx
Key Takeaways
17α-alkylated steroids are hepatotoxic due to their molecular structure, not administration route.
Sublingual and buccal routes do not meaningfully reduce liver toxicity despite bypassing first-pass metabolism.
Injectable 17α-alkylated steroids still expose the liver repeatedly via systemic circulation.
Liver damage arises from androgen receptor activation causing mitochondrial ROS, not just liver metabolism.
Early symptoms of liver toxicity include fatigue, nausea, and elevated liver enzymes; advanced cases show jaundice and tumors.
Monitoring liver enzymes (ALT, AST, GGT) and clinical signs is essential for all users of 17α-alkylated steroids.
Risk mitigation includes limiting cycle duration, avoiding alcohol, and considering hepatoprotective strategies.
The misconception that injectable 17α-alkylated steroids are “liver-safe” can lead to dangerous dosing practices.
Informed decision-making and education are critical to prevent serious liver complications.
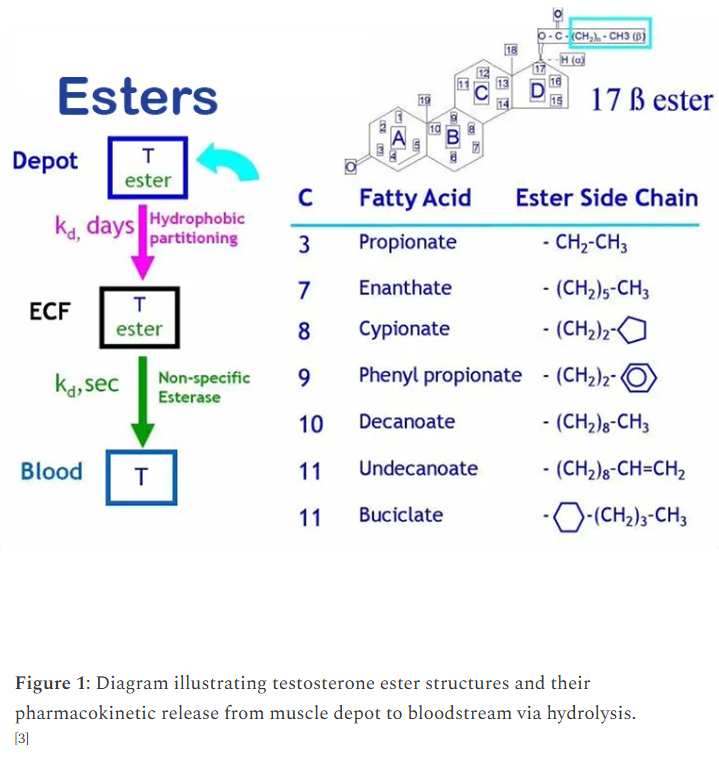
The common wisdom, which is 100% correct and true, is that anabolic-androgenic steroids designed to be taken by intramuscular injection with C-17 esterification – like testosterone (e.g., enanthate), nandrolone (Deca durabolin®, NPP), drostanolone (Masteron), and even trenbolone – are significantly less toxic to the liver (hepatotoxic) than their 17α-alkylated counterparts (17AAs) that are designed to be taken by mouth, like methyltestosterone (METHITEST™, norethandrolone (Nilevar®), methasterone (Superdrol), and mibolerone (R1881; methyltrienolone). [1] C-17 esterification is a chemical modification where an ester like enanthate or decanoate is attached to the steroid to increase its half-life by attaching a carbon chain in the 17-β orientation. [1-1] This modification effectively increases the steroid’s half-life by increasing the residence time inside the depot that is formed in the muscle tissue, from where it slowly dissipates into the blood circulation, where esterases actively hydrolyse the molecule, breaking down those fatty acid bonds of the carbon chain, activating the parent hormone, whose biological half-life is much shorter than the prohormone’s. [1-2] [2]
Esters: Fatty acid bonds of different aliphatic or other carbon chain lengths attached to the steroid’s 17β-hydroxyl group to produce a prodrug for oil vehicle that when injected deep into the belly of muscle is released from depot at a rate determined by its partitioning coefficient according to the ester’s hydrophobicity or water-solubility before entering the extracellular fluid of whole blood where it is rapidly hydrolyzed by esterase…
The myth that many bodybuilders mistakenly embrace, though, is a belief that that this hepatotoxicity is dictated by how they’re taken. By corollary, these same bodybuilders might mistakenly believe that sublingual (or buccal) ingestion of 17AA meaningfully improves their otherwise toxic effects because it bypasses first pass metabolism by the liver.
First Pass vs. Second Pass Metabolism
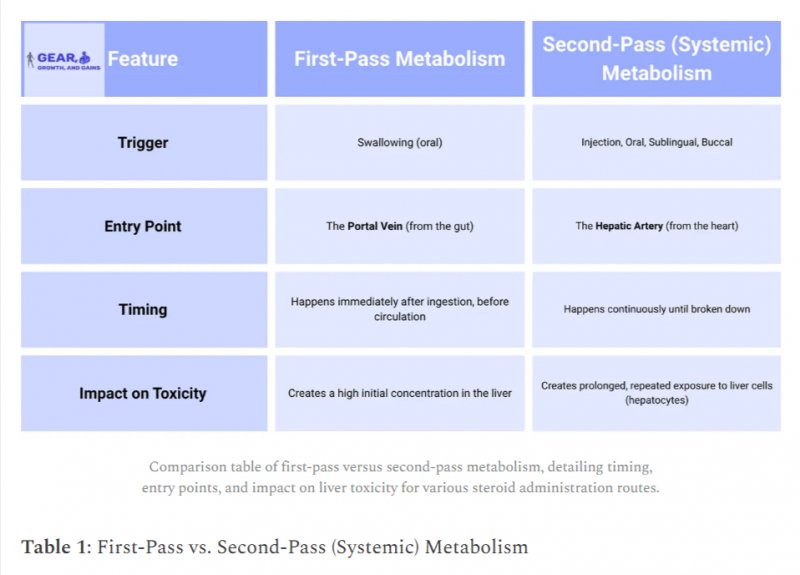
First pass metabolism refers to the process by which a drug is metabolized by the liver before it enters systemic circulation. This can result in a decreased concentration of the drug reaching the bloodstream, as some of it is metabolized and excreted before reaching the intended target site. [4]
Second pass metabolism, on the other hand, refers to the metabolism of a drug that has already entered systemic circulation and is circulating throughout the body. This can occur in various tissues and organs in addition to the liver. [4-1]
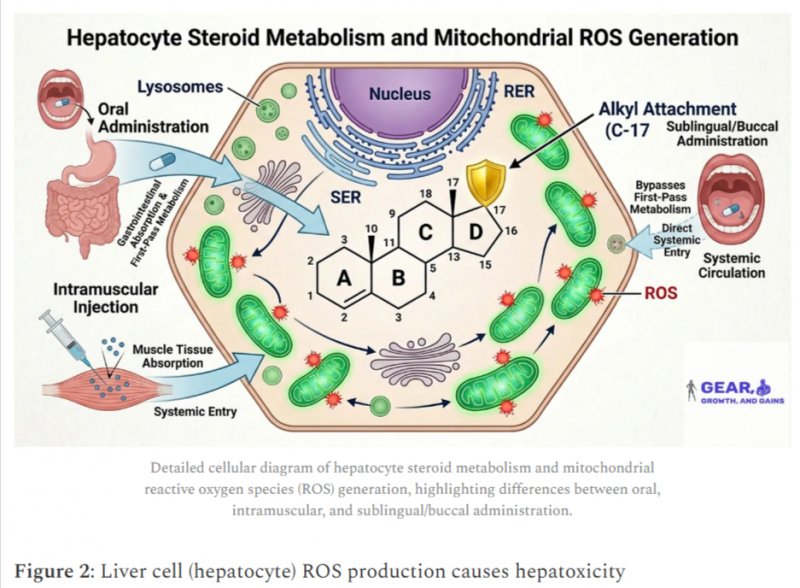
According to this hypothesis, the 17α-alkylated steroids influence mitochondrial function in hepatocytes, specifically affecting how fatty acids are oxidized for energy production. [5-3] The increased CPT1 activity leads to enhanced fatty acid β-oxidation, which in turn generates elevated levels of ROS as metabolic byproducts. [5-4] This pathway provides a biochemical explanation for why the hepatotoxic potential is inherent to the molecular structure itself, independent of how the steroid enters the systemic circulation. [5-5]
This mechanism suggests that the hepatotoxic effects are intrinsically linked to the steroid’s interaction with cellular energy metabolism. The practical implication is profound: if the liver damage results from mitochondrial dysfunction and ROS generation triggered by androgen receptor activation, then the route of administration is practically irrelevant to hepatotoxicity.
Cumulative Exposure Considerations
The total hepatic exposure to a 17α-alkylated steroid depends on multiple factors including dose, duration (cycle length), frequency (how often), half-life and potency (which 17AA). While oral administration may create higher initial liver concentrations, this pass is just a “blip,” an inconsequential surge of drug exposure, since it is the long half-lives (many hours vs. few seconds) that really characterize the 17AAs.
FAQ
People Also Ask
Q: Can sublingual or buccal administration reduce liver toxicity?
A: No. While these routes bypass first-pass metabolism, they do not prevent systemic circulation of the steroid, which continues to expose the liver to toxic effects via androgen receptor activation and mitochondrial ROS generation.
The C-17 Alkylation: A Double-Edged Modification
The 17α-alkylation of anabolic steroids enhances their oral bioavailability by protecting the steroid molecule from rapid hepatic metabolism.
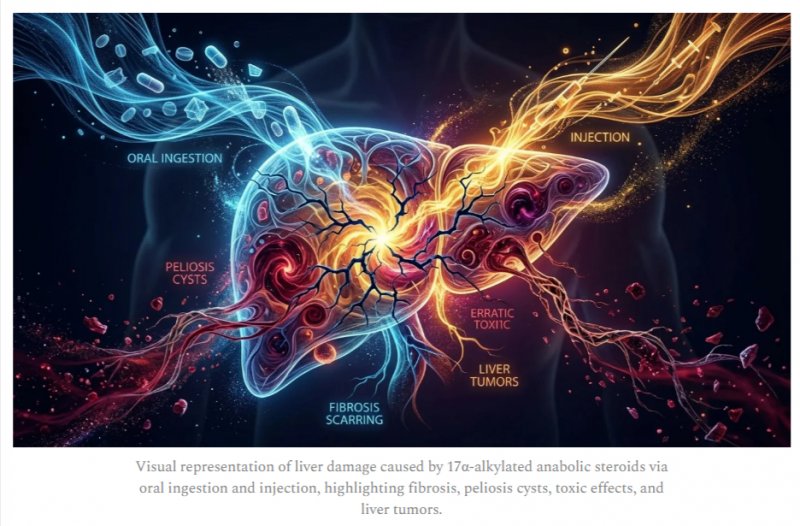
But this same modification which makes oral administration viable also increases hepatotoxicity. The 17α-alkylated variants of anabolic steroids are specifically associated with significant liver toxicity, including serious conditions such as cholestatic icterus and the potential development of liver tumors:
Hepatotoxicity: 17AAs can cause liver damage, primarily thought to result from androgen receptor activation leading to the production of ROS. [6]
Elevated Liver Enzymes: Use often leads to elevations in serum markers including aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and gamma-glutamyl transpeptidase (GGT). [6-1]
Cholestatic Symptoms: In rare cases, toxicity can manifest as jaundice (yellowing of the skin) and pruritus (itching). [6-2]
Peliosis Hepatis: A condition characterized by blood-filled cysts in the liver. [6-3]
Hepatic Tumors: Literature has documented associations with hepatocellular carcinoma and adenoma. [6-4]
Hemorrhage: There are reports of spontaneous haemorrhage of hepatic adenomas in AAS users. [6-5]
This C-17 alkylation, this structural change, is what fundamentally drives hepatotoxicity. The critical point is that this structural modification persists regardless of whether the steroid is taken orally, injected intramuscularly, or administered sublingually.
FAQ
People Also Ask
Q: How can users minimize liver damage risk when using 17α-alkylated steroids?
A: Limit cycle duration, avoid alcohol, maintain hydration, monitor liver enzymes regularly, and consider hepatoprotective strategies. Education and awareness about inherent hepatotoxicity are crucial.
Why Do 17AAs Still Cause Liver Damage Even if Not Swallowed?
The question of why 17AAs cause liver damage no matter the route persists. Let’s break it down.
Liver Blood Flow and Systemic Circulation
The liver receives approximately 25% of cardiac output, with a blood flow rate of roughly 1 liter per minute. [1-3] This massive blood flow ensures that any systemically circulating compound will have substantial and repeated hepatic exposure. When you inject a 17α-alkylated steroid intramuscularly, it enters the bloodstream and must pass through the liver multiple times as blood circulates throughout the body. [4-4]
The pharmacokinetic properties of 17α-alkylated steroids further illuminate why route of administration may not significantly affect hepatotoxic potency. These compounds typically have half-lives in the order of hours with a relatively low volume of distribution (V d). Coupled with a high half-life means that liver cells experience prolonged exposure to these drugs regardless of administration route.
By Type-IIx
Key Takeaways
17α-alkylated steroids are hepatotoxic due to their molecular structure, not administration route.
Sublingual and buccal routes do not meaningfully reduce liver toxicity despite bypassing first-pass metabolism.
Injectable 17α-alkylated steroids still expose the liver repeatedly via systemic circulation.
Liver damage arises from androgen receptor activation causing mitochondrial ROS, not just liver metabolism.
Early symptoms of liver toxicity include fatigue, nausea, and elevated liver enzymes; advanced cases show jaundice and tumors.
Monitoring liver enzymes (ALT, AST, GGT) and clinical signs is essential for all users of 17α-alkylated steroids.
Risk mitigation includes limiting cycle duration, avoiding alcohol, and considering hepatoprotective strategies.
The misconception that injectable 17α-alkylated steroids are “liver-safe” can lead to dangerous dosing practices.
Informed decision-making and education are critical to prevent serious liver complications.
The common wisdom, which is 100% correct and true, is that anabolic-androgenic steroids designed to be taken by intramuscular injection with C-17 esterification – like testosterone (e.g., enanthate), nandrolone (Deca durabolin®, NPP), drostanolone (Masteron), and even trenbolone – are significantly less toxic to the liver (hepatotoxic) than their 17α-alkylated counterparts (17AAs) that are designed to be taken by mouth, like methyltestosterone (METHITEST™, norethandrolone (Nilevar®), methasterone (Superdrol), and mibolerone (R1881; methyltrienolone). [1] C-17 esterification is a chemical modification where an ester like enanthate or decanoate is attached to the steroid to increase its half-life by attaching a carbon chain in the 17-β orientation. [1-1] This modification effectively increases the steroid’s half-life by increasing the residence time inside the depot that is formed in the muscle tissue, from where it slowly dissipates into the blood circulation, where esterases actively hydrolyse the molecule, breaking down those fatty acid bonds of the carbon chain, activating the parent hormone, whose biological half-life is much shorter than the prohormone’s. [1-2] [2]
Esters: Fatty acid bonds of different aliphatic or other carbon chain lengths attached to the steroid’s 17β-hydroxyl group to produce a prodrug for oil vehicle that when injected deep into the belly of muscle is released from depot at a rate determined by its partitioning coefficient according to the ester’s hydrophobicity or water-solubility before entering the extracellular fluid of whole blood where it is rapidly hydrolyzed by esterase…
The myth that many bodybuilders mistakenly embrace, though, is a belief that that this hepatotoxicity is dictated by how they’re taken. By corollary, these same bodybuilders might mistakenly believe that sublingual (or buccal) ingestion of 17AA meaningfully improves their otherwise toxic effects because it bypasses first pass metabolism by the liver.
First Pass vs. Second Pass Metabolism
First pass metabolism refers to the process by which a drug is metabolized by the liver before it enters systemic circulation. This can result in a decreased concentration of the drug reaching the bloodstream, as some of it is metabolized and excreted before reaching the intended target site. [4]
Second pass metabolism, on the other hand, refers to the metabolism of a drug that has already entered systemic circulation and is circulating throughout the body. This can occur in various tissues and organs in addition to the liver. [4-1]
According to this hypothesis, the 17α-alkylated steroids influence mitochondrial function in hepatocytes, specifically affecting how fatty acids are oxidized for energy production. [5-3] The increased CPT1 activity leads to enhanced fatty acid β-oxidation, which in turn generates elevated levels of ROS as metabolic byproducts. [5-4] This pathway provides a biochemical explanation for why the hepatotoxic potential is inherent to the molecular structure itself, independent of how the steroid enters the systemic circulation. [5-5]
This mechanism suggests that the hepatotoxic effects are intrinsically linked to the steroid’s interaction with cellular energy metabolism. The practical implication is profound: if the liver damage results from mitochondrial dysfunction and ROS generation triggered by androgen receptor activation, then the route of administration is practically irrelevant to hepatotoxicity.
Cumulative Exposure Considerations
The total hepatic exposure to a 17α-alkylated steroid depends on multiple factors including dose, duration (cycle length), frequency (how often), half-life and potency (which 17AA). While oral administration may create higher initial liver concentrations, this pass is just a “blip,” an inconsequential surge of drug exposure, since it is the long half-lives (many hours vs. few seconds) that really characterize the 17AAs.
FAQ
People Also Ask
Q: Can sublingual or buccal administration reduce liver toxicity?
A: No. While these routes bypass first-pass metabolism, they do not prevent systemic circulation of the steroid, which continues to expose the liver to toxic effects via androgen receptor activation and mitochondrial ROS generation.
The C-17 Alkylation: A Double-Edged Modification
The 17α-alkylation of anabolic steroids enhances their oral bioavailability by protecting the steroid molecule from rapid hepatic metabolism.
But this same modification which makes oral administration viable also increases hepatotoxicity. The 17α-alkylated variants of anabolic steroids are specifically associated with significant liver toxicity, including serious conditions such as cholestatic icterus and the potential development of liver tumors:
Hepatotoxicity: 17AAs can cause liver damage, primarily thought to result from androgen receptor activation leading to the production of ROS. [6]
Elevated Liver Enzymes: Use often leads to elevations in serum markers including aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and gamma-glutamyl transpeptidase (GGT). [6-1]
Cholestatic Symptoms: In rare cases, toxicity can manifest as jaundice (yellowing of the skin) and pruritus (itching). [6-2]
Peliosis Hepatis: A condition characterized by blood-filled cysts in the liver. [6-3]
Hepatic Tumors: Literature has documented associations with hepatocellular carcinoma and adenoma. [6-4]
Hemorrhage: There are reports of spontaneous haemorrhage of hepatic adenomas in AAS users. [6-5]
This C-17 alkylation, this structural change, is what fundamentally drives hepatotoxicity. The critical point is that this structural modification persists regardless of whether the steroid is taken orally, injected intramuscularly, or administered sublingually.
FAQ
People Also Ask
Q: How can users minimize liver damage risk when using 17α-alkylated steroids?
A: Limit cycle duration, avoid alcohol, maintain hydration, monitor liver enzymes regularly, and consider hepatoprotective strategies. Education and awareness about inherent hepatotoxicity are crucial.
Why Do 17AAs Still Cause Liver Damage Even if Not Swallowed?
The question of why 17AAs cause liver damage no matter the route persists. Let’s break it down.
Liver Blood Flow and Systemic Circulation
The liver receives approximately 25% of cardiac output, with a blood flow rate of roughly 1 liter per minute. [1-3] This massive blood flow ensures that any systemically circulating compound will have substantial and repeated hepatic exposure. When you inject a 17α-alkylated steroid intramuscularly, it enters the bloodstream and must pass through the liver multiple times as blood circulates throughout the body. [4-4]
The pharmacokinetic properties of 17α-alkylated steroids further illuminate why route of administration may not significantly affect hepatotoxic potency. These compounds typically have half-lives in the order of hours with a relatively low volume of distribution (V d). Coupled with a high half-life means that liver cells experience prolonged exposure to these drugs regardless of administration route.
