Bionixx
MuscleHead
- Dec 6, 2011
- 326
- 9
It is with great concern that I have witnessed the increasing use of this compound by bodybuilders and recreational athletes. I have received many emails with questions regarding the efficacy and safety of this substance. This phenolic compound is not a supplement , not even an approved drug for human use. You really want to know what we're talking about?
When we look at the structure of this chemical compound, its simplicity almost makes unbelievable the amount of problems inherent in its use. We are talking about a 6-carbon aromatic ring (benzene) with NO 2 groups at the respective carbons 2 and 4 and also a hydroxyl (OH) group. Sounds simple enough, right? However in physiological chemistry a simple compound is not necessarily a innocuous compound. In the case of DNP, innocuity is what seems to be less ...
Before proceeding to a brief approach to DNP, let me clarify the following:
I do not mind answering emails with questions about whatever compound is of pharmacological origin or just a food supplement. Note, however, that when I give my opinion, people have two hypotheses: they ignore or follow my advice. If you do not want to follow my advice, fine, just ignore what I say. I will not be at all upset that they do, after all it is your body and not mine. What bothers me is that they argue with websites and other often unreliable sources, in an attempt to change my mind. If they look for my "blessing" to make silly they do not send e-mail, since I am not a priest (with all the respect I have for religion). Of course, this "tug of ears" will trigger even more emails in this sense, however here is the clarification / outburst.
Returning to 2,4-dinitrophenol (DNP) ...
It is rumored that several "Gurus" preparers in the world of professional bodybuilding use this compound as a secret weapon to obtain truly defined athletes. I do not know if they use it or not, but even if they do, these individuals have years if not decades of experience in using this substance and the like. Not that this gives them any kind of technical competence to work with this (or other substances), but gives them a much stronger empirical / practical base than an ordinary person.
The only being who can in my perspective aspire to consider himself "Guru" is the Kangaroo, and most likely will be only for his friends. There are no "Gurus" in Bodybuilding (and most probably will not exist in other areas). It's just my opinion...
The explosive history of DNP ...
It is recognized that the use of DNP (C 6 H 4 N 2 O 5 ) leads to a drastic reduction of body weight, while causing an extensive list of side effects (1). From what is known the first death caused by accidental exposure to this compound happened in 1918 (2).
DNP began to be manufactured by the French in the production of ammunition during the First World War, precisely because of its explosive characteristics (3,4). As I said earlier, at this time little would be known about the effects of this compound on weight loss. It was only in 1933 at Stanford University that these potent effects would be confirmed (5).
I once heard one of these "Gurus" (I mean those who are not marsupial mammals) say: DNP is a bomb! He just added: "It was literally explosive and it's still in the right conditions!"
The compound was immediately put on sale for the purpose of aiding in weight-loss diets and was not even subject to a prescription. It is obvious that DNP has the capacity to increase the basal metabolic rate (4,5), leading to a higher energy expenditure (6). Losses of about 1.5 kg / week of body weight are reported under the effect of this compound (7).
The increase in the basal metabolic rate seems to vary among individuals who ingest it, but it is estimated that around 11% per 100 mg of DNP ingested on a regular basis (8-10). As they are expected to calculate in the 1930s, this drug seemed to be the solution to an emerging obesity (the same thing happened with the discovery of leptin more recently). However, side effects increased progressively, especially with the appearance of cataracts, which would lead to the suspension of its use by the Federal Food, Drug and Cosmetic Act of 1938 (1,11).
With the suspension of its sale for medical purposes, obviously the number of fatal cases and adverse reactions declined. Nevertheless, some fatal cases were reported, even after the suspension of its commercialization (12,13). It is also believed that the compound was given to Russian soldiers during World War II as a way to protect them against low temperatures (14). If we look at the graph below, we can easily see that since the beginning of the millennium, fatal cases have been increasing.
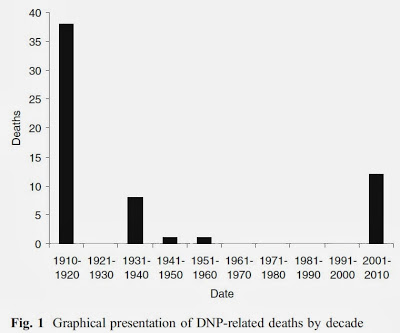
The main concern I have about this drug is that it is sold easily on the internet . The amount of kids who embark on this dangerous and sometimes fatal game is very large. Many are underage who, by influence of "A" or "B" decide, in an offhand way, to order this product online as if it were a simple supplement. It is common with the use of this drug to reach 38.9 ° C. Look at the table below (summary of fatal cases), I think the data speaks for itself.
Mechanism of action
These phenol derivatives generally tend to cause hyperthermia, tachycardia, diaphoresis and tachypnea. Next, I will briefly discuss the respective mechanisms involved in weight loss and toxicity.
Decoupling function of oxidative phosphorylation
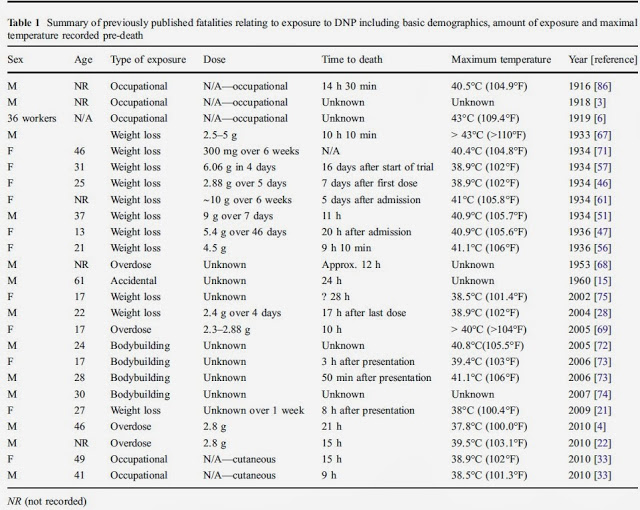
DNP decreases the formation of phosphate bonds in the mitochondria at the same time as it stimulates the systemic consumption of oxygen (15). This effect is described biochemically by its decoupling function in the formation of adenosine triphosphate (ATP).
ATP is the end product of the Krebs cycle as well as CO 2 and H 2 O. During glycolysis there is a positive balance of 2 ATP molecules, however the vast majority of ATP molecules (in a possible total of 36-38) is produced during the final phase of oxidative phosphorylation.
At this stage the ATP synthase converts the ADP to ATP by the addition of an inorganic phosphate (Pi) molecule. What DNP does is to prevent the uptake of inorganic phosphate molecules by mitochondria (16,17). The result is inhibition of mitochondrial energy processes as well as excessive accumulation of extra-mitochondrial Pi (18). DNP is also an ionophore molecule, since it promotes the proton (H + ) exit by the mitochondrial membrane (10).
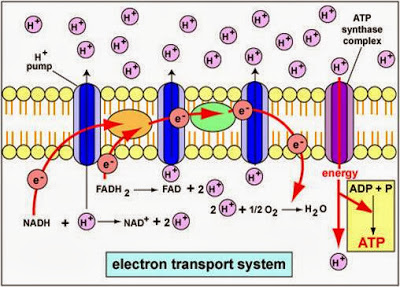
This imbalance in the electrochemical gradient leads to the energy dissipating in the form of heat instead of forming ATP, increasing the energy expenditure (kcal). This is the situation responsible for the homeostatic deregulation of the temperature, which in turn leads to the respective appearance of hyperthermia pictures (19).
If it escaped on the table above, some patients reached temperatures around 43 ° C. So if you are going to use this drug and you do not know what you are doing, think first: Well or badly spent?
Stimulating glycolysis
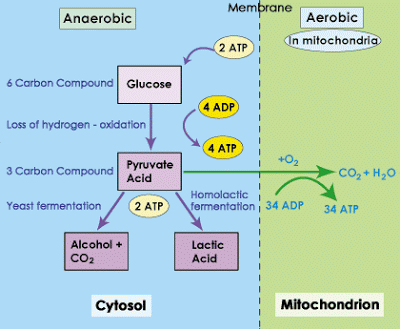
The glycolytic effect of DNP is most likely associated with its effect on muscle contraction (20). The consumption of carbohydrates increases exponentially with the administration of this drug (18). The pyruvic acid to be converted into CO 2 and H 2 O needs to be followed by the aerobic pathway, since oxidative phosphorylation is reduced by the compound, the anaerobic pathway becomes preferential leading to a higher production of lactic acid and consequent acidosis (16,18,21).
Accumulation of phosphate and potassium
DNP increases potassium (K + ) concentration (22) leading to hyperkalemia toxicity (23). The contribution of phosphate to toxicity is still unclear and requires further investigation (17,24).
Teratogenic and carcinogenic effect
In guinea pigs this compound has shown teratogenic effects (changes in embryonic morphology) and carcinogenic effects (25).
Luckily on the days that run it is becoming less common accidental DNP poisoning. The most common intoxication is usually by its ingestion with weight loss goals. There are two drugs that really bother me when used improperly by both recreational and professional athletes: DNP and insulin.
My opinion is clear, use of drugs for aesthetic or sports purposes definitely NO! With regard to the two substances mentioned above, when used abusively and uncontrolled lead to only a shortcut: Death.
The position I have in relation to these two drugs does not come only from the literature. I've met a lot of athletes who got real scares with these substances. I do not want with this article to pass on the idea that I am a purist in this matter of pharmacological ergogenic substances, none of that, I want you to realize that with certain things you do not really play!
References:
1. Colman E. Dinitrophenol and obesity: an early twentieth-century regulatory dilemma. Regul. Toxicol. Pharmacol. 2007 Jul; 48 (2): 115-7.
2. Warthin AS. A fatal case of toxic jaundice caused by dinitrophenol. Bull Int Assoc Med Mus. 1918; 7: 123-6.
3. Perkins RG. A study of munitions intoxications in France. Pub Heal. Rep. 1919; 34: 2335.
4. CUTTING WC. ACTIONS AND USES OF DINITROPHENOL. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Jul 15; 101 (3): 193-5.
5. TAINTER ML. USE OF DINITROPHENOL IN OBESITY AND RELATED CONDITIONS. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Nov 4; 101 (19): 1472.
6. TAINTER ML, CUTTING WC, HINES E. EFFECTS OF MODERATE DOSES OF DINITROPHENOL ON THE ENERGY EXCHANGE AND NITROGEN METABOLISM OF PATIENTS UNDER CONDITIONS OF RESTRICTED DIETARY. J. Pharmacol. Exp. Ther. 1935 Nov 1; 55 (3): 326-53.
7. Grundlingh J, Dargan PI, El-Zanfaly M, Wood DM. 2,4-dinitrophenol (DNP): a weight loss agent with significant acute toxicity and risk of death. J. Med. Toxicol. 2011 Sep; 7 (3): 205-12.
8. Dunlop DM. THE USE OF 2: 4-DINITROPHENOL AS A METABOLIC STIMULANT. Br. Med. J. 1934 Mar 24; 1 (3820): 524-7.
9. Tainter ML. TREATMENT OF ACUTE DINITROPHENOL POISONING. JAMA J. Am. Med. Assoc. American Medical Association; 1935 Mar 30; 104 (13): 1071.
10. Harper JA, Dickinson K, Brand MD. Mitochondrial uncoupling as a target for drug development for the treatment of obesity. Obes. Rev. 2001 Nov; 2 (4): 255-65.
11. ATSDR. Toxicological Profile: Dinitrophenols [Internet]. 1995 [cited 2013 Oct 12]. Available from: http://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=729&tid=132
12. GISCLARD JB, WOODWARD MM. 2,4-Dinitrophenol poisoning; a case report. J. Ind. Hyg. Toxicol. 1946 Mar; 28: 47-51.
13. CANN HM, VERHULST HL. Fatality from acute dinitrophenol derivative poisoning. Am. J. Dis. Child. American Medical Association; 1960 Dec; 100: 947-8.
14. Kurt TL, Anderson R, Petty C, Bost R, Reed G, Holland J. Dinitrophenol in weight loss: the poison center and public health safety. Vet. Hum. Toxicol. 1986 Dec; 28 (6): 574-5.
15. Unknown. DINITROPHENOL AND ACCELERATED TISSUE METABOLISM. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Dec 30; 101 (27): 2122.
16. Rognstad R, Katz J. The effect of 2,4-dinitrophenol on adipose-tissue metabolism. Biochem. J. 1969 Feb; 111 (4): 431-44.
17. Issekutz B. Effect of propranolol in dinitrophenol poisoning. Arch. Int. Pharmacodyn. The R. 1984 Dec; 272 (2): 310-9.
18. SIMON EW. MECHANISMS OF DINITROPHENOL TOXICITY. Biol. Rev. 1953 Nov; 28 (4): 453-78.
19. Hoch FL, Hogan FP. Hyperthermia, muscle rigidity, and uncoupling in skeletal muscle mitochondria in rats treated with halothane and 2,4-dinitrophenol. Anesthesiology. 1973 Mar; 38 (3): 237-43.
20. El-Guindy MM, Neder AC, Gomes CB. 2,4-Dinitrophenol - mechanism of action. Cell. Mol Biol. Incl. Cyto. Enzymol. 1981 Jan; 27 (5): 399-402.
21. Krahl ME, Clowes GHA. SOME EFFECTS OF DINITROCRESOL ON OXIDATION AND FERMENTATION. J. Biol. Chem. 1935 Oct 1; 111 (2): 355-69.
22. MUDGE GH. Electrolyte and water metabolism of rabbit kidney slices; effect of metabolic inhibitors. Am. J. Physiol. 1951 Oct; 167 (1): 206-23.
23. Jiang Jiukun, Yuan Zhihua, Huang Weidong, Wang Jiezan. 2, 4-dinitrophenol poisoning caused by non-oral exposure. Toxicol. Ind. Health. 2011 May; 27 (4): 323-7.
24. Moffatt EJ, Miyamoto MD. Effect of sodium and calcium channel blockade on the increase in spontaneous transmitter release produced by the mitochondrial inhibitor, dinitrophenol. J. Pharmacol. Exp. Ther. 1988 Feb; 244 (2): 613-8.
25. Takahashi M, Sunaga M, Hirata-Koizumi M, Hirose A, Kamata E, Ema M. Reproductive and developmental toxicity screening study of 2,4-dinitrophenol in rats. Environ. Toxicol. 2009 Feb; 24 (1): 74-81.
When we look at the structure of this chemical compound, its simplicity almost makes unbelievable the amount of problems inherent in its use. We are talking about a 6-carbon aromatic ring (benzene) with NO 2 groups at the respective carbons 2 and 4 and also a hydroxyl (OH) group. Sounds simple enough, right? However in physiological chemistry a simple compound is not necessarily a innocuous compound. In the case of DNP, innocuity is what seems to be less ...
Before proceeding to a brief approach to DNP, let me clarify the following:
I do not mind answering emails with questions about whatever compound is of pharmacological origin or just a food supplement. Note, however, that when I give my opinion, people have two hypotheses: they ignore or follow my advice. If you do not want to follow my advice, fine, just ignore what I say. I will not be at all upset that they do, after all it is your body and not mine. What bothers me is that they argue with websites and other often unreliable sources, in an attempt to change my mind. If they look for my "blessing" to make silly they do not send e-mail, since I am not a priest (with all the respect I have for religion). Of course, this "tug of ears" will trigger even more emails in this sense, however here is the clarification / outburst.
Returning to 2,4-dinitrophenol (DNP) ...
It is rumored that several "Gurus" preparers in the world of professional bodybuilding use this compound as a secret weapon to obtain truly defined athletes. I do not know if they use it or not, but even if they do, these individuals have years if not decades of experience in using this substance and the like. Not that this gives them any kind of technical competence to work with this (or other substances), but gives them a much stronger empirical / practical base than an ordinary person.
The only being who can in my perspective aspire to consider himself "Guru" is the Kangaroo, and most likely will be only for his friends. There are no "Gurus" in Bodybuilding (and most probably will not exist in other areas). It's just my opinion...
The explosive history of DNP ...
It is recognized that the use of DNP (C 6 H 4 N 2 O 5 ) leads to a drastic reduction of body weight, while causing an extensive list of side effects (1). From what is known the first death caused by accidental exposure to this compound happened in 1918 (2).
DNP began to be manufactured by the French in the production of ammunition during the First World War, precisely because of its explosive characteristics (3,4). As I said earlier, at this time little would be known about the effects of this compound on weight loss. It was only in 1933 at Stanford University that these potent effects would be confirmed (5).
I once heard one of these "Gurus" (I mean those who are not marsupial mammals) say: DNP is a bomb! He just added: "It was literally explosive and it's still in the right conditions!"
The compound was immediately put on sale for the purpose of aiding in weight-loss diets and was not even subject to a prescription. It is obvious that DNP has the capacity to increase the basal metabolic rate (4,5), leading to a higher energy expenditure (6). Losses of about 1.5 kg / week of body weight are reported under the effect of this compound (7).
The increase in the basal metabolic rate seems to vary among individuals who ingest it, but it is estimated that around 11% per 100 mg of DNP ingested on a regular basis (8-10). As they are expected to calculate in the 1930s, this drug seemed to be the solution to an emerging obesity (the same thing happened with the discovery of leptin more recently). However, side effects increased progressively, especially with the appearance of cataracts, which would lead to the suspension of its use by the Federal Food, Drug and Cosmetic Act of 1938 (1,11).
With the suspension of its sale for medical purposes, obviously the number of fatal cases and adverse reactions declined. Nevertheless, some fatal cases were reported, even after the suspension of its commercialization (12,13). It is also believed that the compound was given to Russian soldiers during World War II as a way to protect them against low temperatures (14). If we look at the graph below, we can easily see that since the beginning of the millennium, fatal cases have been increasing.
The main concern I have about this drug is that it is sold easily on the internet . The amount of kids who embark on this dangerous and sometimes fatal game is very large. Many are underage who, by influence of "A" or "B" decide, in an offhand way, to order this product online as if it were a simple supplement. It is common with the use of this drug to reach 38.9 ° C. Look at the table below (summary of fatal cases), I think the data speaks for itself.
Mechanism of action
These phenol derivatives generally tend to cause hyperthermia, tachycardia, diaphoresis and tachypnea. Next, I will briefly discuss the respective mechanisms involved in weight loss and toxicity.
Decoupling function of oxidative phosphorylation
DNP decreases the formation of phosphate bonds in the mitochondria at the same time as it stimulates the systemic consumption of oxygen (15). This effect is described biochemically by its decoupling function in the formation of adenosine triphosphate (ATP).
ATP is the end product of the Krebs cycle as well as CO 2 and H 2 O. During glycolysis there is a positive balance of 2 ATP molecules, however the vast majority of ATP molecules (in a possible total of 36-38) is produced during the final phase of oxidative phosphorylation.
At this stage the ATP synthase converts the ADP to ATP by the addition of an inorganic phosphate (Pi) molecule. What DNP does is to prevent the uptake of inorganic phosphate molecules by mitochondria (16,17). The result is inhibition of mitochondrial energy processes as well as excessive accumulation of extra-mitochondrial Pi (18). DNP is also an ionophore molecule, since it promotes the proton (H + ) exit by the mitochondrial membrane (10).
This imbalance in the electrochemical gradient leads to the energy dissipating in the form of heat instead of forming ATP, increasing the energy expenditure (kcal). This is the situation responsible for the homeostatic deregulation of the temperature, which in turn leads to the respective appearance of hyperthermia pictures (19).
If it escaped on the table above, some patients reached temperatures around 43 ° C. So if you are going to use this drug and you do not know what you are doing, think first: Well or badly spent?
Stimulating glycolysis
The glycolytic effect of DNP is most likely associated with its effect on muscle contraction (20). The consumption of carbohydrates increases exponentially with the administration of this drug (18). The pyruvic acid to be converted into CO 2 and H 2 O needs to be followed by the aerobic pathway, since oxidative phosphorylation is reduced by the compound, the anaerobic pathway becomes preferential leading to a higher production of lactic acid and consequent acidosis (16,18,21).
Accumulation of phosphate and potassium
DNP increases potassium (K + ) concentration (22) leading to hyperkalemia toxicity (23). The contribution of phosphate to toxicity is still unclear and requires further investigation (17,24).
Teratogenic and carcinogenic effect
In guinea pigs this compound has shown teratogenic effects (changes in embryonic morphology) and carcinogenic effects (25).
Luckily on the days that run it is becoming less common accidental DNP poisoning. The most common intoxication is usually by its ingestion with weight loss goals. There are two drugs that really bother me when used improperly by both recreational and professional athletes: DNP and insulin.
My opinion is clear, use of drugs for aesthetic or sports purposes definitely NO! With regard to the two substances mentioned above, when used abusively and uncontrolled lead to only a shortcut: Death.
The position I have in relation to these two drugs does not come only from the literature. I've met a lot of athletes who got real scares with these substances. I do not want with this article to pass on the idea that I am a purist in this matter of pharmacological ergogenic substances, none of that, I want you to realize that with certain things you do not really play!
References:
1. Colman E. Dinitrophenol and obesity: an early twentieth-century regulatory dilemma. Regul. Toxicol. Pharmacol. 2007 Jul; 48 (2): 115-7.
2. Warthin AS. A fatal case of toxic jaundice caused by dinitrophenol. Bull Int Assoc Med Mus. 1918; 7: 123-6.
3. Perkins RG. A study of munitions intoxications in France. Pub Heal. Rep. 1919; 34: 2335.
4. CUTTING WC. ACTIONS AND USES OF DINITROPHENOL. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Jul 15; 101 (3): 193-5.
5. TAINTER ML. USE OF DINITROPHENOL IN OBESITY AND RELATED CONDITIONS. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Nov 4; 101 (19): 1472.
6. TAINTER ML, CUTTING WC, HINES E. EFFECTS OF MODERATE DOSES OF DINITROPHENOL ON THE ENERGY EXCHANGE AND NITROGEN METABOLISM OF PATIENTS UNDER CONDITIONS OF RESTRICTED DIETARY. J. Pharmacol. Exp. Ther. 1935 Nov 1; 55 (3): 326-53.
7. Grundlingh J, Dargan PI, El-Zanfaly M, Wood DM. 2,4-dinitrophenol (DNP): a weight loss agent with significant acute toxicity and risk of death. J. Med. Toxicol. 2011 Sep; 7 (3): 205-12.
8. Dunlop DM. THE USE OF 2: 4-DINITROPHENOL AS A METABOLIC STIMULANT. Br. Med. J. 1934 Mar 24; 1 (3820): 524-7.
9. Tainter ML. TREATMENT OF ACUTE DINITROPHENOL POISONING. JAMA J. Am. Med. Assoc. American Medical Association; 1935 Mar 30; 104 (13): 1071.
10. Harper JA, Dickinson K, Brand MD. Mitochondrial uncoupling as a target for drug development for the treatment of obesity. Obes. Rev. 2001 Nov; 2 (4): 255-65.
11. ATSDR. Toxicological Profile: Dinitrophenols [Internet]. 1995 [cited 2013 Oct 12]. Available from: http://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=729&tid=132
12. GISCLARD JB, WOODWARD MM. 2,4-Dinitrophenol poisoning; a case report. J. Ind. Hyg. Toxicol. 1946 Mar; 28: 47-51.
13. CANN HM, VERHULST HL. Fatality from acute dinitrophenol derivative poisoning. Am. J. Dis. Child. American Medical Association; 1960 Dec; 100: 947-8.
14. Kurt TL, Anderson R, Petty C, Bost R, Reed G, Holland J. Dinitrophenol in weight loss: the poison center and public health safety. Vet. Hum. Toxicol. 1986 Dec; 28 (6): 574-5.
15. Unknown. DINITROPHENOL AND ACCELERATED TISSUE METABOLISM. JAMA J. Am. Med. Assoc. American Medical Association; 1933 Dec 30; 101 (27): 2122.
16. Rognstad R, Katz J. The effect of 2,4-dinitrophenol on adipose-tissue metabolism. Biochem. J. 1969 Feb; 111 (4): 431-44.
17. Issekutz B. Effect of propranolol in dinitrophenol poisoning. Arch. Int. Pharmacodyn. The R. 1984 Dec; 272 (2): 310-9.
18. SIMON EW. MECHANISMS OF DINITROPHENOL TOXICITY. Biol. Rev. 1953 Nov; 28 (4): 453-78.
19. Hoch FL, Hogan FP. Hyperthermia, muscle rigidity, and uncoupling in skeletal muscle mitochondria in rats treated with halothane and 2,4-dinitrophenol. Anesthesiology. 1973 Mar; 38 (3): 237-43.
20. El-Guindy MM, Neder AC, Gomes CB. 2,4-Dinitrophenol - mechanism of action. Cell. Mol Biol. Incl. Cyto. Enzymol. 1981 Jan; 27 (5): 399-402.
21. Krahl ME, Clowes GHA. SOME EFFECTS OF DINITROCRESOL ON OXIDATION AND FERMENTATION. J. Biol. Chem. 1935 Oct 1; 111 (2): 355-69.
22. MUDGE GH. Electrolyte and water metabolism of rabbit kidney slices; effect of metabolic inhibitors. Am. J. Physiol. 1951 Oct; 167 (1): 206-23.
23. Jiang Jiukun, Yuan Zhihua, Huang Weidong, Wang Jiezan. 2, 4-dinitrophenol poisoning caused by non-oral exposure. Toxicol. Ind. Health. 2011 May; 27 (4): 323-7.
24. Moffatt EJ, Miyamoto MD. Effect of sodium and calcium channel blockade on the increase in spontaneous transmitter release produced by the mitochondrial inhibitor, dinitrophenol. J. Pharmacol. Exp. Ther. 1988 Feb; 244 (2): 613-8.
25. Takahashi M, Sunaga M, Hirata-Koizumi M, Hirose A, Kamata E, Ema M. Reproductive and developmental toxicity screening study of 2,4-dinitrophenol in rats. Environ. Toxicol. 2009 Feb; 24 (1): 74-81.
