You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
IronSoul
TID Board Of Directors
- Apr 2, 2013
- 6,334
- 2,106
ECA stack?
While both EPHEDRINE and CAFFEINE increase the metabolic rate and have anoretic effects, WHY is ASPIRIN being used?
jim
I've been wondering this a lot lately dr Jim. I understand the synergistic effect staking ephedrine and caffeine but unsure of the role aspirin plays. I hope to hear something back on this one
C
CBS
Senior Member
- Jan 7, 2014
- 183
- 59
ECA stack?
While both EPHEDRINE and CAFFEINE increase the metabolic rate and have anoretic effects, WHY is ASPIRIN being used?
jim
I've been wondering this a lot lately dr Jim. I understand the synergistic effect staking ephedrine and caffeine but unsure of the role aspirin plays. I hope to hear something back on this one
I believe the idea came from a couple of studies on ECA's effect on post-prandial thermogenesis, however, the addition of aspirin only had a small beneficial effect on weight loss in obese subjects. If I remember correctly, the purported mechanism was that aspirin inhibits prostaglandins which prevent the release of adrenaline and noradrenaline, and when combined with caffeine, it supposedly has a synergistic effect that enhances thermogenesis and metabolism by sustaining elevated cAMP levels.
The studies that showed a benefit for aspirin needed around 300 mg tid. Obviously a lot. EC works fine on its own, especially if already lean.
CBS
Mini Forklift Ⓥ
The Veganator
- Dec 23, 2012
- 4,313
- 730
I presume it's role would be as a blood-thinner, but I could be wrong.
Thanks for that CBS ~ looks like I was wrong lol.I believe the idea came from a couple of studies on ECA's effect on post-prandial thermogenesis, however, the addition of aspirin only had a small beneficial effect on weight loss in obese subjects. If I remember correctly, the purported mechanism was that aspirin inhibits prostaglandins which prevent the release of adrenaline and noradrenaline, and when combined with caffeine, it supposedly has a synergistic effect that enhances thermogenesis and metabolism by sustaining elevated cAMP levels.
The studies that showed a benefit for aspirin needed around 300 mg tid. Obviously a lot. EC works fine on its own, especially if already lean.
CBS
C
CBS
Senior Member
- Jan 7, 2014
- 183
- 59
This link offers a good review on the use of ECA. http://members.tripod.com/jpe_sportscience/ephedrine.html
Role of Ephedrine, Caffeine and Aspirin
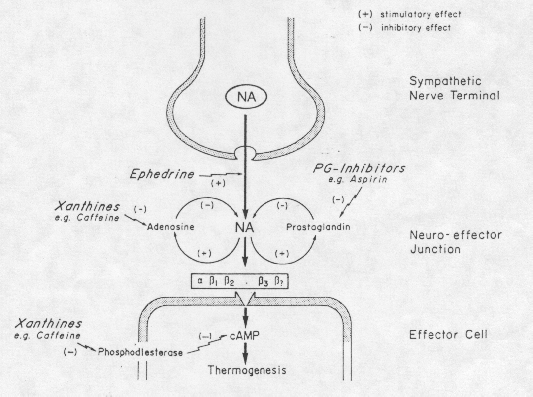
from A. G. Dullo, Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S35-40.
Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S73-8.Ephedrine, caffeine and aspirin: safety and efficacy for treatment of human obesity.
Daly PA[SUP]1[/SUP], Krieger DR, Dulloo AG, Young JB, Landsberg L.
Abstract
The safety and efficacy of a mixture of ephedrine (75-150mg), caffeine (150mg) and aspirin (330mg), in divided premeal doses, were investigated in 24 obese humans (mean BMI 37.0) in a randomized double blind placebo-controlled trial. Energy intake was not restricted. Overall weight loss over 8 weeks was 2.2kg for ECA vs. 0.7 kg for placebo (p < 0.05). 8 of 13 placebo subjects returned 5 months later and received ECA in an unblinded crossover. After 8 weeks, mean weight loss with ECA was 3.2 kg vs 1.3 kg for placebo (p = 0.036). 6 subjects continued on ECA for 7 to 26 months. After 5 months on ECA, average weight loss in 5 of these was 5.2 kg compared to 0.03 kg gained during 5 months between studies with no intervention (p = 0.03). The sixth subject lost 66 kg over 13 months by self-imposed caloric restriction. In all studies, no significant changes in heart rate, blood pressure, blood glucose, insulin, and cholesterol levels, and no differences in the frequency of side effects were found. ECA in these doses is thus well tolerated in otherwise healthy obese subjects, and supports modest, sustained weight loss even without prescribed caloric restriction, and may be more effective in conjunction with restriction of energy intake.
Am J Clin Nutr. 1987 Mar;45(3):564-9.Aspirin as a promoter of ephedrine-induced thermogenesis: potential use in the treatment of obesity.
Dulloo AG, Miller DS.
Abstract
Chronic administration of aspirin to obese mice had no effect on energy balance and body composition. In contrast, ephedrine increased energy expenditure by 9% and reduced body weight and body fat by 18% and 50%, respectively: obesity, however, was reduced but not reversed. In the presence of both ephedrine and aspirin, increase in energy expenditure found during treatment with ephedrine alone was doubled, and the obese group lost greater than 75% of body fat: obesity was reversed. These studies indicate that although aspirin administered alone has no influence on energy balance it can markedly potentiate thermogenic properties of ephedrine, effects which led to a normalization of body composition of the obese to that of the lean. Such ephedrine-aspirin mixtures, often found in over-the-counter preparations for asthma and bronchial disorders, could be put to new use as aids for treatment of human obesity.
Role of Ephedrine, Caffeine and Aspirin

from A. G. Dullo, Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S35-40.
Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S73-8.Ephedrine, caffeine and aspirin: safety and efficacy for treatment of human obesity.
Daly PA[SUP]1[/SUP], Krieger DR, Dulloo AG, Young JB, Landsberg L.
Abstract
The safety and efficacy of a mixture of ephedrine (75-150mg), caffeine (150mg) and aspirin (330mg), in divided premeal doses, were investigated in 24 obese humans (mean BMI 37.0) in a randomized double blind placebo-controlled trial. Energy intake was not restricted. Overall weight loss over 8 weeks was 2.2kg for ECA vs. 0.7 kg for placebo (p < 0.05). 8 of 13 placebo subjects returned 5 months later and received ECA in an unblinded crossover. After 8 weeks, mean weight loss with ECA was 3.2 kg vs 1.3 kg for placebo (p = 0.036). 6 subjects continued on ECA for 7 to 26 months. After 5 months on ECA, average weight loss in 5 of these was 5.2 kg compared to 0.03 kg gained during 5 months between studies with no intervention (p = 0.03). The sixth subject lost 66 kg over 13 months by self-imposed caloric restriction. In all studies, no significant changes in heart rate, blood pressure, blood glucose, insulin, and cholesterol levels, and no differences in the frequency of side effects were found. ECA in these doses is thus well tolerated in otherwise healthy obese subjects, and supports modest, sustained weight loss even without prescribed caloric restriction, and may be more effective in conjunction with restriction of energy intake.
Am J Clin Nutr. 1987 Mar;45(3):564-9.Aspirin as a promoter of ephedrine-induced thermogenesis: potential use in the treatment of obesity.
Dulloo AG, Miller DS.
Abstract
Chronic administration of aspirin to obese mice had no effect on energy balance and body composition. In contrast, ephedrine increased energy expenditure by 9% and reduced body weight and body fat by 18% and 50%, respectively: obesity, however, was reduced but not reversed. In the presence of both ephedrine and aspirin, increase in energy expenditure found during treatment with ephedrine alone was doubled, and the obese group lost greater than 75% of body fat: obesity was reversed. These studies indicate that although aspirin administered alone has no influence on energy balance it can markedly potentiate thermogenic properties of ephedrine, effects which led to a normalization of body composition of the obese to that of the lean. Such ephedrine-aspirin mixtures, often found in over-the-counter preparations for asthma and bronchial disorders, could be put to new use as aids for treatment of human obesity.
dr jim
MuscleHead
- Apr 7, 2014
- 785
- 168
Thanks CBS BUT the authors didn't compare the EC and the ECA groups to determine if ASA made any difference in thermogenesis and considering the negative impact ANY NSAID may have on protein synthesis (theoretically) I'M NOT BUYING IT.
The only mechanism I'm aware of regarding ASA and "weight loss" involves the uncoupling of oxidative phosphorlation (much like DNP) which increases thermogenesis in VERY HIGH DOSES!
Agreed MATE drop the ASA
Best
JIM
The only mechanism I'm aware of regarding ASA and "weight loss" involves the uncoupling of oxidative phosphorlation (much like DNP) which increases thermogenesis in VERY HIGH DOSES!
Agreed MATE drop the ASA
Best
JIM
Last edited:
IronSoul
TID Board Of Directors
- Apr 2, 2013
- 6,334
- 2,106
Hey Dr. Jim, for a bronkaid caffeine stack, what dosage of the bronkaid would you recommend. I just started today and only using one tab bronkaid (25mg ephedrine) and one 200mg caffeine tab. I will possibly run that dose split evenly throughout the day for 3 times but also just considering the one time pre workout.
C
CBS
Senior Member
- Jan 7, 2014
- 183
- 59
Thanks CBS BUT the authors didn't compare the EC and the ECA groups to determine if ASA made any difference in thermogenesis and considering the negative impact ANY NSAID may have on protein synthesis (theoretically) I'M NOT BUYING IT.
The only mechanism I'm aware of regarding ASA and "weight loss" involves the uncoupling of oxidative phosphorlation (much like DNP) which increases thermogenesis in VERY HIGH DOSES!
Agreed MATE drop the ASA
Best
JIM
I'm not trying to make a case for it - just reporting the supposed mechanism behind it, speculative as it may be.
There is at least one study that compared ECA with EC but I've been unable to locate it. But just going from memory, the benefit seen was small and only occurred in obese subjects. Lean subjects got no benefit. For that reason, plus the increased risk of GI effects from using aspirin at 300mg tid for weeks on end, it should be avoided, IMO.
CBS
C
CBS
Senior Member
- Jan 7, 2014
- 183
- 59
Hey Dr. Jim, for a bronkaid caffeine stack, what dosage of the bronkaid would you recommend. I just started today and only using one tab bronkaid (25mg ephedrine) and one 200mg caffeine tab. I will possibly run that dose split evenly throughout the day for 3 times but also just considering the one time pre workout.
The doses used in most studies were 20mg ephedrine and 200mg caffeine tid. I have no idea about pre-workout - perhaps Jim can advise on that one.
Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S69-72.Safety and efficacy of long-term treatment with ephedrine, caffeine and an ephedrine/caffeine mixture.
Toubro S[SUP]1[/SUP], Astrup AV, Breum L, Quaade F.
Abstract
In a randomized, placebo-controlled, double blind study, 180 obese patients were treated by diet (4.2 MJ/day) and either an ephedrine/caffeine combination (20mg/200mg), ephedrine (20mg), caffeine (200mg) or placebo 3 times a day for 24 weeks. 141 patients completed this part of the study. All medication was stopped between week 24-26 in order to catch any withdrawal symptoms. From week 26 to 50, 99 patients completed treatment with the ephedrine/caffeine compound in an open trial design, resulting in a statistically significant (p = 0.02) weight loss of 1.1kg. In another randomized, double-blind, placebo-controlled 8 week study on obese subjects we found the mentioned compound showed lean body mass conserving properties. We conclude that the ephedrine/caffeine combination is effective in improving and maintaining weight loss, further it has lean body mass saving properties. The side effects are minor and transient and no withdrawal symptoms have been found.
Int J Obes Relat Metab Disord. 1993 Dec;17 Suppl 3:S73-7; discussion S82.The acute and chronic effects of ephedrine/caffeine mixtures on energy expenditure and glucose metabolism in humans.
Toubro S[SUP]1[/SUP], Astrup A, Breum L, Quaade F.
Abstract
This paper describes a 24-week open follow-up trial with reduced obese patients all receiving an ephedrine/caffeine combination (20 mg/200 mg) three times a day. The study was a continuation of a previous 24-week double-blind placebo-controlled study where the ephedrine/caffeine mixture had shown superior weight-reducing properties when compared with either ephedrine alone (20 mg) or caffeine alone (200 mg) three times a day. The medication was stopped between weeks 24-26 in order to evaluate withdrawal symptoms. The follow-up period was from weeks 26 to 50. Of 127 patients included, 99 completed the follow-up treatment, which resulted in an additional weight loss of 1.1 kg (P = 0.02). Adverse drug reactions were all minor and temporary. We conclude that the ephedrine/caffeine combination is safe and effective in long-term treatment in improving and maintaining weight loss. The side-effects are minor and transient and no clinically relevant withdrawal symptoms have been observed.
IronSoul
TID Board Of Directors
- Apr 2, 2013
- 6,334
- 2,106
The doses used in most studies were 20mg ephedrine and 200mg caffeine tid. I have no idea about pre-workout - perhaps Jim can advise on that one.
Int J Obes Relat Metab Disord. 1993 Feb;17 Suppl 1:S69-72.Safety and efficacy of long-term treatment with ephedrine, caffeine and an ephedrine/caffeine mixture.
Toubro S[SUP]1[/SUP], Astrup AV, Breum L, Quaade F.
Abstract
In a randomized, placebo-controlled, double blind study, 180 obese patients were treated by diet (4.2 MJ/day) and either an ephedrine/caffeine combination (20mg/200mg), ephedrine (20mg), caffeine (200mg) or placebo 3 times a day for 24 weeks. 141 patients completed this part of the study. All medication was stopped between week 24-26 in order to catch any withdrawal symptoms. From week 26 to 50, 99 patients completed treatment with the ephedrine/caffeine compound in an open trial design, resulting in a statistically significant (p = 0.02) weight loss of 1.1kg. In another randomized, double-blind, placebo-controlled 8 week study on obese subjects we found the mentioned compound showed lean body mass conserving properties. We conclude that the ephedrine/caffeine combination is effective in improving and maintaining weight loss, further it has lean body mass saving properties. The side effects are minor and transient and no withdrawal symptoms have been found.
Int J Obes Relat Metab Disord. 1993 Dec;17 Suppl 3:S73-7; discussion S82.The acute and chronic effects of ephedrine/caffeine mixtures on energy expenditure and glucose metabolism in humans.
Toubro S[SUP]1[/SUP], Astrup A, Breum L, Quaade F.
Abstract
This paper describes a 24-week open follow-up trial with reduced obese patients all receiving an ephedrine/caffeine combination (20 mg/200 mg) three times a day. The study was a continuation of a previous 24-week double-blind placebo-controlled study where the ephedrine/caffeine mixture had shown superior weight-reducing properties when compared with either ephedrine alone (20 mg) or caffeine alone (200 mg) three times a day. The medication was stopped between weeks 24-26 in order to evaluate withdrawal symptoms. The follow-up period was from weeks 26 to 50. Of 127 patients included, 99 completed the follow-up treatment, which resulted in an additional weight loss of 1.1 kg (P = 0.02). Adverse drug reactions were all minor and temporary. We conclude that the ephedrine/caffeine combination is safe and effective in long-term treatment in improving and maintaining weight loss. The side-effects are minor and transient and no clinically relevant withdrawal symptoms have been observed.
Thanks cbs, that's about what I planned on doing. But I'll be 5mg over at each dose. Loved it today
